Korea ARPA-H Project
ARPA-H is a Korea-led national research and development initiative that aims to overcome unmet diseases, including rare diseases, by advancing large-scale research and therapeutic development.
Korea ARPA-H Project
ARPA-H is a Korean national research and development project dedicated to overcoming unmet medical needs, including rare diseases, through large-scale research and therapeutic development.
Seoul National University Hospital (SNUH) has been selected as the lead institution for a research project under the Korean ARPA-H Project in 2025 and is advancing the initiative titled
“Development of a Personalized Innovative Treatment Platform for Pediatric Rare Diseases and N-of-1 Clinical Trials.”
The “Personalized Innovative Treatment Platform for Pediatric Rare Disease Patients and N-of-1 Clinical Trials Project” aims to improve survival rates and quality of life for children with rare diseases by developing a patient-specific treatment platform based on innovative technologies.
Although advances in precision medicine and genomic medicine, along with strengthened institutional frameworks, have improved diagnostic rates for rare diseases, most patients still face limited access to effective treatments after diagnosis.
In particular, for severe pediatric rare diseases, conventional drug development approaches typically require many years, resulting in significantly limited treatment accessibility.
To overcome these challenges, approaches that analyze individual genetic variants and design personalized therapies have recently been introduced worldwide.
The SNUH consortium is launching a full-scale research initiative to design and manufacture individualized ASO (antisense oligonucleotide)-based therapies for patients with rare diseases.
ASO technology is a precision medicine strategy known to target the fundamental molecular mechanisms of disease by binding to mRNA transcribed from specific genes and regulating protein production.
This project includes the development and administration of customized ASO therapeutics for individual patients through N-of-1 clinical trials, representing the first such initiative of its kind in Korea.
Through this effort, the project aims to establish a new gene therapy–based clinical platform for children with severe rare diseases and to build a foundation for expansion to patient groups with diverse genetic variants.
Ultimately, this project is expected to contribute to expanding treatment opportunities for patients with rare diseases in Korea.
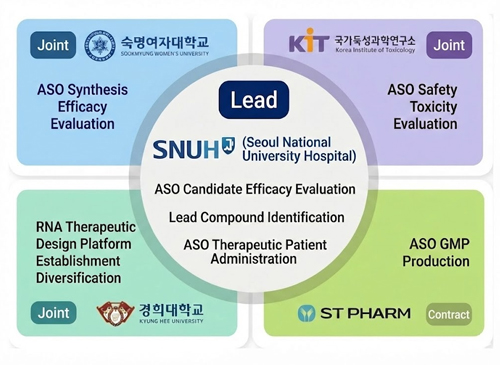
ASO-Based Personalized Therapy Research for Rare Diseases
The Seoul National University Hospital Consortium is advancing the development of ASO-based personalized therapeutics for rare disease patients, along with N-of-1 clinical trials, through the Korean ARPA-H (ARPA-H) project.
* ASO (Antisense Oligonucleotide):
A synthetic oligonucleotide therapeutic that binds to specific mRNA molecules to inhibit protein translation or regulate gene expression.
